Ponceau S staining (preparation and protocol)
Introduction
Ponceau S (a.k.a Acid Red 112) is a red colour a diazo dye used for reversible staining of proteins in Western blot. It was first by O Salinovich and R C Montelaroused in 1986 as an alternative for Coomassie brilliant blue staining[1].
Ponceau is one of the many dyes used for staining of proteins. These dyes have different binding properties and detection limits. The table below (adapted from Goldman et al., 2016[2]) gives an overview of detection limit and usability on different types of membranes.
| Stain | Minimum amount detected | Membrane type | ||
|---|---|---|---|---|
| PVDF | Nitrocellulose | Nylon | ||
| Amido black | 50 ng | + | + | + |
| Coomassie blue | 50 ng | + | − | + |
| Ponceau S | 200 ng | + | + | * |
| Colloidal gold | 2 ng | + | + | − |
| Colloidal silver | 5 ng | + | + | − |
| India ink | 5 ng | + | + | − |
| MemCode | 25 ng | + | + | − |
Table 1. Comparision of different membranes and compatibility with stains.
*Nylon membranes may not be useful as the dye binds strongly to the membrane and gives high background.
Although the Ponceau S detecting ability is low, it is one of the most used stains due to its reversibility of staining and low price compared to colloids. Due to the reversible staining (without deleterious effects on proteins) the samples can be used for other downstream applications such as immunoblotting and sequencing.
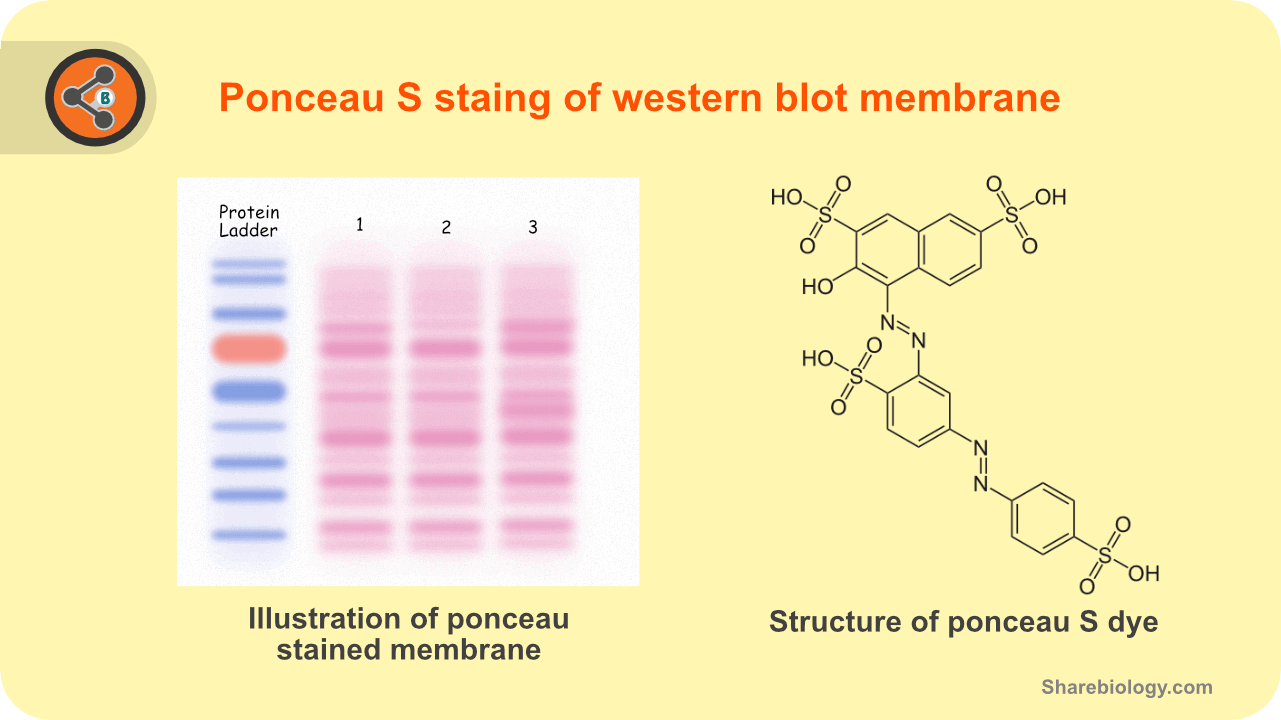
Figure 1.Illustration depicting the ponceau S staining in western blot (left) and structure of ponceau S stain.
Applications
- The most common use is Western blots as a stain for proteins to detect and record the pattern of protein bands.
- It can also be used for protein quantification spectrophotometrically [3].
Composition
| Reagent | Molecular weight | 1X molarity | Add for 500ml | Add for 1L |
|---|---|---|---|---|
| Ponceau S | 760.57 (4 Na salt) | 1.3 mM (0.1 %) | 0.5 g | 1 g |
| Acetic acid (glacial) | 60.05 | 0.874 M (5 %) | 25 ml | 50 ml |
Table 1. Composition of ponceau S stain.
The standard composition is 0,1 % of dye and 5% of acetic acid (Biotium, Novus biologicals and Sigma-Aldrich).
there are several formulations available with a difference in the percentage of acetic acid and dye. Some formulations also use trichloroacetic acid and sulfosalicylic acid as a replacement to acetic acid.
According to Sander et al., 2019, the staining method works well regardless of the concentration of Ponceau S (2%-0.001%), acetic acid (1%-20%), or other acid types[4]. Therefore, a more dilute solution can also be used 0.01 % of dye and acetic acid (1%).
We are providing other types of recipes used at the end of the post. You can use those recipes in case if the above-mentioned recipe fails.
pH
The standard pH of this solution is acidic thanks to acetic acid. No need to adjust.
Preparation
- For the preparing 500 ml of ponceau S staining solution, add 25 ml of glacial acetic acid to 400 ml of distilled water.
- Add 0.5 gm of ponceau S tetrasodium salt to the acetic acid prepared above.
- Stir the mixture to dissolve.
- Make up the solution to 500 ml using distilled water.
Protocol for ponceau S staining
- Remove the membrane from the blotting chamber.
- Wash the membrane with TBS-T.
- Add the Ponceau staining solution till the membrane submerges.
- Keep it for gentle ration for 5-10 min at room temperature.
- Remove the staining solution (can be re-used) and add distilled water to remove the background.
- A prolonged period of distilled water washing cause destaining of protein ban
Protocol for ponceau S destaining
Ponceau-stained membrane can be detained using:
Distilled water.
5% acetic acid solution.
0.1M NaOH solution.
Storage
Store at room temperature or 4°C. Do not freeze.
Points to be noted
Glacial acid is a concentrated corrosive acid. Therefore, work with care – use gloves and safety glasses.
Other recipes
CSHL
0.5% (w/v) Ponceau S
1% acetic acid
Reference: CSHL
Wallert and Prvost lab
Ponceau S: 0.1% (w/v)
Acetic acid: 1% (v/v)
Reference: Wallert and Provost Lab
Sigma-Aldrich
2% Ponceau S (w/v)
30% TCA,
30% sulfosalicylic acid
Reference: Sigma-Aldrich
Reference
- Reversible Staining and Peptide Mapping of Proteins Transferred to Nitrocellulose After Separation by Sodium Dodecylsulfate-Polyacrylamide Gel Electrophoresis.
- Detection of Proteins on Blot Membranes.
- Measuring Protein Concentration on Nitrocellulose and After the Electrophoretic Transfer of Protein to Nitrocellulose.
- Ponceau S waste: Ponceau S staining for total protein normalization.
- Biotium.
- Novus biologicals.
- Sigma-Aldrich.
